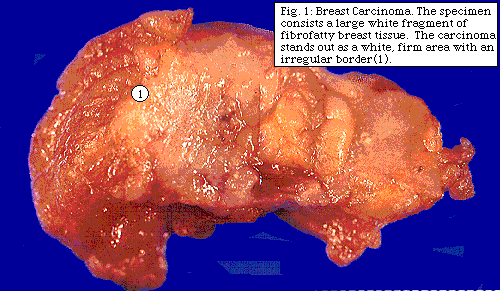
Author: Colleen M. Powell, M.D., Assistant Professor of Pathology and Laboratory Medicine, University of Pennsylvania Medical Center
BREAST BIOPSY
Breast lesions suspected
of being malignant are usually evaluated first by biopsy. Palpable (large enough
to feel) masses can be directly excised. Sometimes, however, the lesion is detected
only by mammography. In these cases the radiologist places a needle to guide the
surgeon's excision. The needle, which remains in place, also identifies the radiographically
abnormal area for the pathologist. These excisions are performed in the operating
room.
Diagnostic tissue can also be obtained using "needle biopsies" or "fine needle aspirations". These are less invasive procedures than excision and can be performed in the radiology department or the doctors office. "Needle biopsies" use a thin needle to obtain a small core of tissue which is sent to surgical pathology. An accurately performed needle biopsy produces tissue that is felt to be representative of the lesion. It is not an attempt to completely remove the mass or abnormality. "Fine needle aspiration" (FNA) involves a similar procedure. With this method, cells from the suspicious area are aspirated and placed on a glass slides. This material is evaluated in cytopathology.
Both needle biopsies and fine needle aspirations are followed by an excisional biopsy if (1) cancer is present that needs to be removed or (2) the tissue core or aspirated cells are either non-diagnostic or suspicious for cancer and the clinician feels that further evaluation is indicated.
Breast tissue is evaluated
immediately in surgical pathology. First the pathologist describes the tissue
and correlates what she sees with any accompanying radiographs. If the biopsy
was performed for suspicious areas on mammography, it is determined that the
lesion is present in the tissue removed. 
The pathologist then applies ink to the external surface of the specimen. This is to identify the "surgical margin" or the plane cut by the surgeon. The specimen is then sectioned to examine all areas.
When a grossly evident lesion is suspicious for malignancy, (see figure to right) a frozen section is performed. If cancer is found, and tissue is sufficient, a portion is then taken for special studies. (See "special studies")
After the frozen section the entire specimen is placed in formalin for fixation and preparation of glass slides for microscopic review and definitive diagnosis. Small biopsies are processed entirely. Larger specimens are generously sampled with special attention to visible lesions, fibrous or more firm areas and margins. All tissue adjacent to localizing needles is also evaluated microscopically.
When the microscopic slides are examined for the final diagnosis, the pathologist pays special attention to certain details. These are: tumor size, type and grade, size of invasive component, lymphatic or vascular invasion, extent of the in-situ component, surgical margins, lymph nodes (when available) and background changes. Each of these issues will be discussed separately below.
The breast contains acini and ducts necessary for the production and secretion of milk. The acini are clustered together in lobules. The ducts and lobules are supported by stroma consisting of fibrous tissue and fat.
Malignant cells may be confined to the ducts and acini (lobules). In these cases the tumor is called "in-situ" meaning that it has remained where it began and has not invaded the supporting stroma. In-situ carcinomas are divided into (1) "ductal-carcinoma- in-situ" or "intraductal carcinoma" and (2) "lobular-carcinoma-in situ".
Intraductal carcinoma is considered a pre-invasive lesion; given time many would progess to invasive carcinoma. These lesions need to be completely excised. (see "Surgical margins" and "Extent of intraductal carcinoma").
Lobular-carcinoma-in
situ is not considered pre-invasive but is felt to be a strong predictor
of future new breast carcinoma in either the same or the opposite breast. For
these reason, while it is important to document the presence of lobular-carcinoma-in-situ,
it is not currently considered necessary for it to be completely excised.
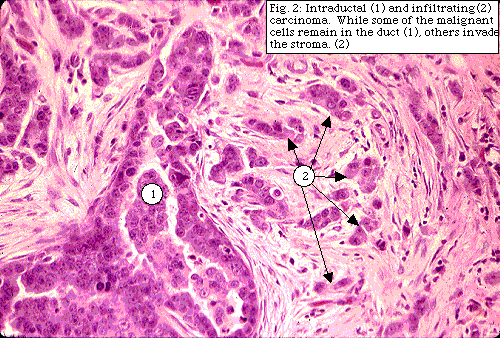
Malignant cells that have moved out of the duct or acini (lobule) into the stroma are called "invasive" or "infiltrating" mammary carcinomas. Infiltrating (invasive) tumors are divided into
Malignant cells able to escape the duct or lobule are more aggressive and capable of spreading beyond the breast to adjacent lymph nodes and distant sites (ie metastasize).
"Breast cancer" can include any combination of in-situ and infiltrating carcinoma.
Tumor size, recorded at the time of frozen section examination, is included in the final pathologic diagnosis. Some tumors, too small to be appreciated in the whole specimen, are only seen microscopically. In these cases, the size of the tumor is measured from the glass slide.
Although some tumors are composed entirely of infiltrating or invasive tumor cells, many are a combination of in-situ and infiltrating carcinoma. In these tumors the percentage of intraductal carcinoma is estimated. (See "Extent of intraductal carcinoma").
Tumors which are predominantly intraductal with only small areas of invasion are called "microinvasive". The microinvasive areas are described and measured on the glass slide if possible.
The size of the invasive
component, as well as whole tumor size, is important to assess prognosis and
direct therapy. Generally tumors with smaller areas of invasion do better.
TUMOR GRADE
The appearance of the malignant
cells and the pattern of their invasion into the stroma varies between tumors.
Tumors that most closely recapitulate the normal tissue are considered to be
"low-grade" (well-differentiated). Tumors that bear little resemblance to the
tissue they arose from are called "high-grade" (poorly differentiated) Some
tumors represent an "intermediate grade" (moderately differentiated) between
the other two extremes. 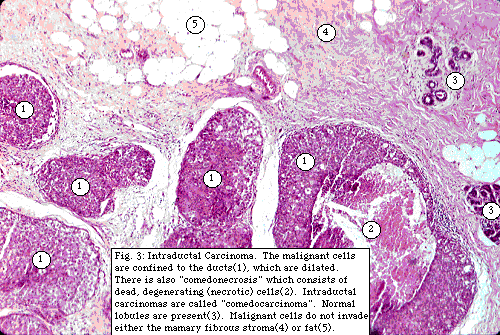
Intraductal carcinoma is subclassified according to its pattern of growth into
The pathologist documents the presence or absence of tumor cells (tumor emboli) in lymphatic or vascular channels . The presence of "tumor emboli" signify increased risk of (1) subsequent distant metastases and (2) local recurrence in patients treated with breast conservation therapy.
The surgical pathologist
evaluates the resection margin identified by the black ink applied to the original
specimen. The presence or absence of intraductal carcinoma or infiltrating tumor
(ductal or lobular) at the margin is documented. Tumor at the surgical margin
raises the concern that the primary tumor may not have been completely removed.
Some tumors need to be re-excised to make sure the margins are free of tumor.
Other tumors may be managed with appropriate adjustments of the scheduled radiation
therapy.
EXTENT OF INTRADUCTAL CARCINOMA
It has been shown that breast cancers with an extensive intraductal component are more likely to recur after conservative breast therapy. The pathologist documents the presence or absence of extensive intraductal malignancy. If the main tumor contains significant intraductal carcinoma and intraductal carcinoma is seen in the surrounding breast tissue, then "extensive intraductal carcinoma" is present.
Because it is important in prognosis and therapy planning, the pathologist also documents whether the malignancy is confined to one area or is seen in multiple foci.
Axillary lymph nodes are usually removed in patients with invasive breast carcinoma. The pathologist examines the lymph nodes for metastatic tumor. The total number of lymph nodes recovered and the number containing tumor are documented. Generally patients are stratified as (1) no tumor seen (2) 1-3 affected nodes (3) 4-9 affected nodes and (4) more than ten affected nodes. It is also noted whether tumor is present in the fibrous-fatty tissue around the lymph nodes or if multiple lymph nodes are matted together cancer. Treatment options are partly based on the number of involved lymph nodes (called "nodal status")
The benign changes in the
breast are also described. "Fibrocystic changes" include the formation of cysts,
increased 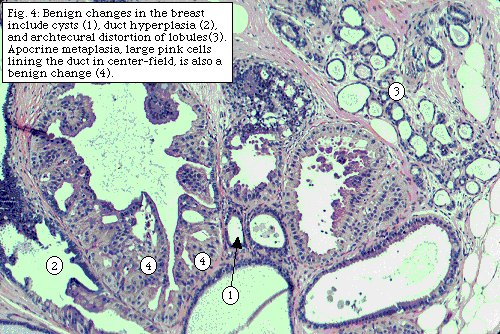 fibrous
tissue, benign patterns of duct or lobule distortion, and benign cellular changes.
(Figure to the right). The
cells lining ducts or acini (lobules) may show increased growth without evidence
of malignancy. This change is called "duct hyperplasia" (or "papillomatosis").
Sometimes the cells in duct hyperplasia are atypical (abnormal but not cancerous).
Significant duct hyperplasia, especially if atypia is present, places the patient
at an increased risk of subsequent breast carcinoma. Benign hyperplasia can
also be seen in the acini (lobules) where it is called "lobular hyperplasia"
or "atypical lobular hyperplasia" if some cellular abnormalities are seen. Atypical
lobular hyperplasia, like lobular-carcinoma-in -situ, is not pre-invasive but
is associated with an increased risk of subsequent breast malignancies.
fibrous
tissue, benign patterns of duct or lobule distortion, and benign cellular changes.
(Figure to the right). The
cells lining ducts or acini (lobules) may show increased growth without evidence
of malignancy. This change is called "duct hyperplasia" (or "papillomatosis").
Sometimes the cells in duct hyperplasia are atypical (abnormal but not cancerous).
Significant duct hyperplasia, especially if atypia is present, places the patient
at an increased risk of subsequent breast carcinoma. Benign hyperplasia can
also be seen in the acini (lobules) where it is called "lobular hyperplasia"
or "atypical lobular hyperplasia" if some cellular abnormalities are seen. Atypical
lobular hyperplasia, like lobular-carcinoma-in -situ, is not pre-invasive but
is associated with an increased risk of subsequent breast malignancies.
Because they serve as a markers of increased risk of subsequent breast cancer, "duct hyperplasia", "atypical duct hyperplasia", "lobular hyperplasia", and "atypical lobular hyperplasia, are documented if present. However, like lobular-carcinoma-in-situ, these changes are not considered pre-invasive and no attempt is currently made to remove all tissue with these changes.
When clinically indicated, the entire breast is removed. There are several types of mastectomy and all of these operations remove the entire breast.
The pathologist evaluates mastectomies similar to biopsies. Ink is applied to the surgical margin and the tissue is serially sectioned and inspected. If there has been a previous biopsy, the surgical incision and biopsy cavity are described. Since the entire specimen cannot be examined microscopically, standard sections are submitted. If present, the biopsy cavity is generously sampled looking for residual tumor. Additional suspicious areas are also taken for microscopic evaluation. Finally, representative sections of each of the four breast quadrants, skin and nipple are submitted for review. If present, all lymph nodes are also evaluated. Because the entire specimen is not examined microscopically, it is possible that microscopic tumor foci have not been discovered. This is not currently considered to be a problem since the entire organ has been removed.
Small deposits of calcium can be seen on mammography. If their pattern is suspicious for cancer, a needle is placed to direct the surgeon and the pathologist. An xray of the specimen after surgery is usually performed and the radiograph sent to pathology with the biopsy. This allows the pathologist to confirm that the calcifications are present in the excised material. When examining the specimen microscopically, the pathologist notes the presence or absence of the calcifications. Although some calcifications seen on mammography cannot be seen by the pathologist, most are visible. Presence of calcifications in the microscopic sections re-assures the pathologist that she has examined the pertinent areas.
CONTROVERSIES IN BREAST
PATHOLOGY
The practice of pathology is part science and part art. While there is general diagnostic agreement among pathologists, some issues generate debate. Studies have shown that a panel of recognized expert breast pathologists can fail to reach consensus on certain controversial breast lesions.
Both frozen and routine microscopic sections are traditionally stained with a combination of the dyes eosin and hematoxylin which color the cell nuclei blue and the cytoplasm pink. Additional staining methods, focused on specific cellular elements, are used when indicated. "Immunohistochemical" stains utilize specific antibodies to cellular constituents. "Special" stains utilize chemical reactions to impart different colors to molecules of interest.
Traditionally the amount of tumor estrogen and progesterone hormone receptors has been determined using a biochemical test. This method requires fresh tissue obtained at the time of frozen section. Estrogen and progesterone hormone receptors can also be studied with immunohistochemical stains. Immunohistochemistry can use either fresh tissue (cut onto slides at the time of frozen section) or tissue that has been processed and embedded in wax. This method is especially useful for small tumors that were either not large enough to be seen in the original (unprocessed) specimen or too small to spare tissue for special studies. The status of these hormone receptors is used to direct hormonal therapy.
Fresh tissue, if sufficient at the time of initial evaluation, is also obtained for "flow cytometry". Flow cytometry quantifies both the rate of tumor cell growth and the amount of nuclear DNA. This data can help determine prognosis and direct therapy.
Other immunohistochemical markers, performed in experimental protocols, are currently being studied in breast tissue. It is hoped that, in the future, some of these will prove useful in guiding therapy.